|
Regulatory Guidance Emphasizes Disclosure and Warns of Potential RisksĪs highlighted in a recent speech by then-Commissioner and now Acting Chair of the SEC Allison Herren Lee, the SEC has recognized the innovative potential of SPACs to "enliven public markets and expand investor opportunities." Lee, however, also emphasized the need for enhanced scrutiny of disclosures relating to "relevant risks and sponsor compensation," and for exploring whether additional protections are necessary to prevent sponsors from pursuing less-than-ideal acquisitions. In the current market environment, SPAC IPOs and PIPEs have been attracting enormous amounts of capital from investors. In addition, the capital raises effected by private investments in public equity (PIPEs), which often accompany de-SPAC transactions, provide these companies with an additional influx of capital. SPACs have grown in popularity for many reasons, including because there is a perception that they involve an easier process than traditional IPOs in that they provide private companies with a quicker way to go public with more price transparency. A sponsor team, which is often composed of sophisticated business managers and investors, raises these funds for the SPAC and guides it through the IPO and, upon identifying a target, through one or more acquisitions (the so-called de-SPAC transaction). In general, the SPAC is required to use the trust funds within two years to make an acquisition, or it must return the funds to its investors. In the SPAC IPO, the cash proceeds from the offering of common stock and warrants to purchase common stock are placed into a trust account for use to acquire an operating company (the target). A SPAC is a publicly traded, non-operating company that is used solely as a vehicle to acquire an operating target company in the future. SPAC deals have existed in various forms for more than 25 years. This advisory surveys the lay of the land regarding these risks and identifies practical suggestions about ways to anticipate and mitigate them. SPAC market participants-including sponsors, target companies, directors and officers on both sides of the transactions, and investment banks-should be aware of the enforcement and litigation risks involved. We expect this will continue and perhaps increase, particularly if retail investors get involved in SPAC IPOs. 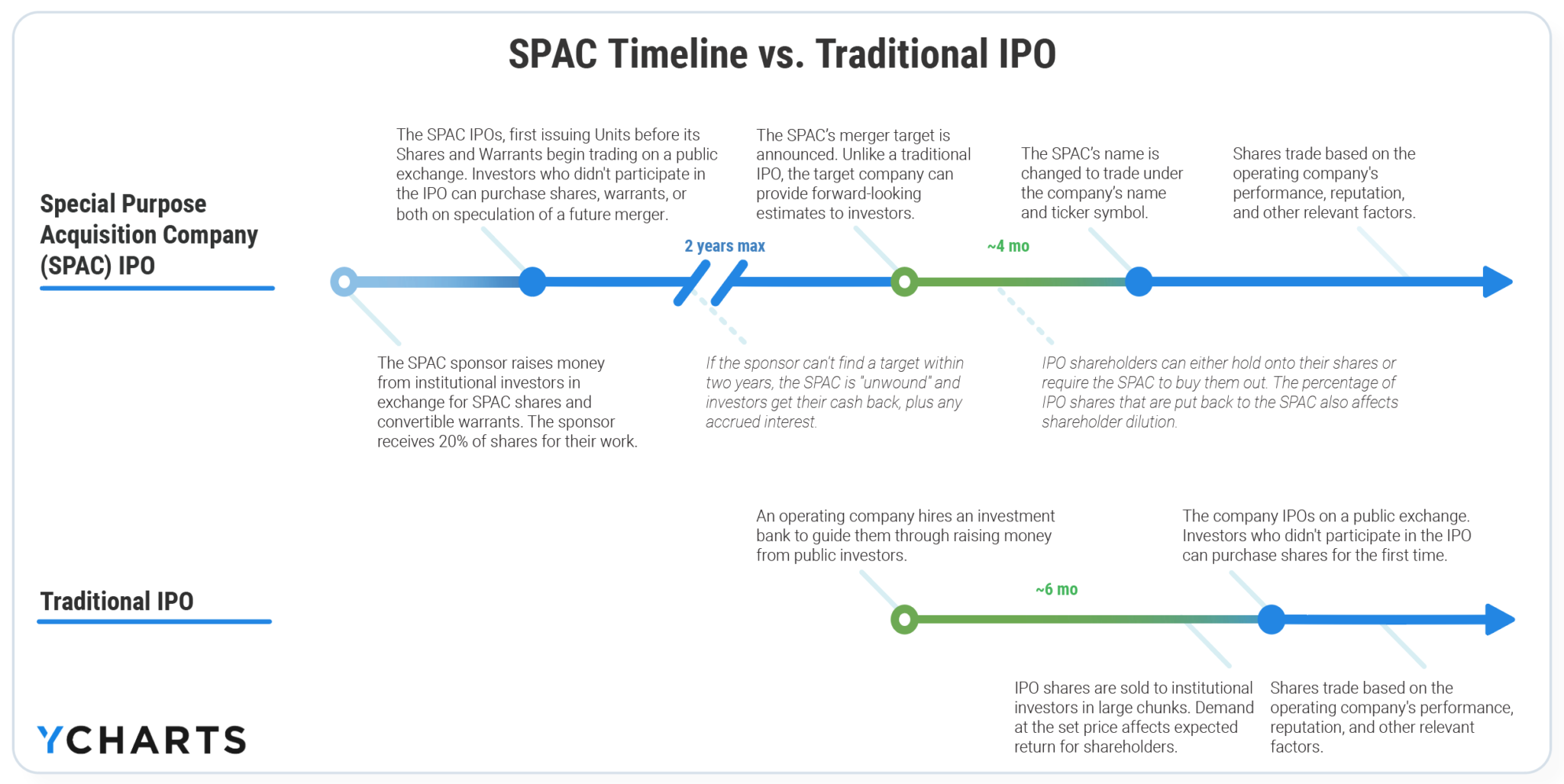
Inevitably, the growth in SPAC deal activity has attracted scrutiny from market regulators, including the US Securities and Exchange Commission (SEC) and Financial Industry Regulatory Authority (FINRA), as well as private plaintiffs. 
In the first three months of 2021, there have been approximately 300 initial public offerings (IPOs) of special purpose acquisition companies (SPACs) that have raised $100 billion, which are significant increases over the record number of transactions and amounts raised in all of 2020. Subscribe to our "Biden-Harris Agenda" mailing list to receive our analyses. With deep insights into policies and policymakers, Arnold & Porter has established the Biden-Harris Agenda Resource Team to advise clients on the changing landscape.
0 Comments

The island of stability is a hypothetical region in the top right cluster of nuclides that contains isotopes far more stable than other transuranic elements.Drip lines have only been established for some elements. Beyond the proton drip line along the upper right, nuclides decay by proton emission.Beyond the neutron drip line along the lower left, nuclides decay by neutron emission.Examples include boron-10, carbon-12, and nitrogen-14 (as N − Z = 0 for each pair), or boron-12, carbon-14, and nitrogen-16 (as N − Z = 2 for each pair). Like isobars, they follow diagonal lines, but at right angles to the isobar lines (from upper-left to lower-right). Isodiaphers are nuclides with the same difference between their numbers of neutrons and protons ( N − Z).Examples include carbon-14, nitrogen-14, and oxygen-14 in the table above. Isobars neighbor each other diagonally from lower-left to upper-right. mass number) but different numbers of protons and neutrons. Isobars are nuclides with the same number of nucleons (i.e.Examples include carbon-14, nitrogen-15, and oxygen-16 in the table above. Isotones neighbor each other horizontally. Isotones are nuclides with the same number of neutrons but differing numbers of protons.Examples include carbon-12, carbon-13, and carbon-14 in the table above. Isotopes are nuclides with the same number of protons but differing numbers of neutrons that is, they have the same atomic number and are therefore the same chemical element. 
The darker more stable isotope region departs from the line of protons (Z) = neutrons (N), as the element number Z becomes larger 
The trends in this section refer to the following chart, which shows Z increasing to the right and N increasing downward, a 90° clockwise rotation of the above landscape-orientation charts. It has become a basic tool of the nuclear community. Today, there are several nuclide charts, four of which have a wide distribution: the Karlsruhe Nuclide Chart, the Strasbourg Universal Nuclide Chart, the Chart of the Nuclides from the Japan Atomic Energy Agency (JAEA), and the Nuclide Chart from Knolls Atomic Power Laboratory in the United States. Its 7th edition was made available in 2006. In 1958, Walter Seelmann-Eggebert and Gerda Pfennig published the first edition of the Karlsruhe Nuclide Chart. This representation was first published by Kurt Guggenheimer in 1934 and expanded by Giorgio Fea in 1935, Emilio Segrè in 1945 or Glenn Seaborg. Nuclide charts organize nuclides along the X axis by their numbers of neutrons and along the Y axis by their numbers of protons, out to the limits of the neutron and proton drip lines. It contrasts with a periodic table, which only maps their chemical behavior, since isotopes (nuclides which are variants of the same element) do not differ chemically to any significant degree, with the exception of hydrogen. Description and utility Ī chart or table of nuclides maps the nuclear, or radioactive, behavior of nuclides, as it distinguishes the isotopes of an element. The chart of the nuclides is also known as the Segrè chart, after the Italian physicist Emilio Segrè. This system of ordering nuclides can offer a greater insight into the characteristics of isotopes than the better-known periodic table, which shows only elements and not their isotopes. Each point plotted on the graph thus represents a nuclide of a known or hypothetical chemical element.
|
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
